Treating Ebola with 'bioresonance'
And cancer. And depression. And Dengue Fever, Japanese Encephalitis, Yellow Fever, hayfever, allergies, fungal infections, Crohn's disease, ADHD, IBS…
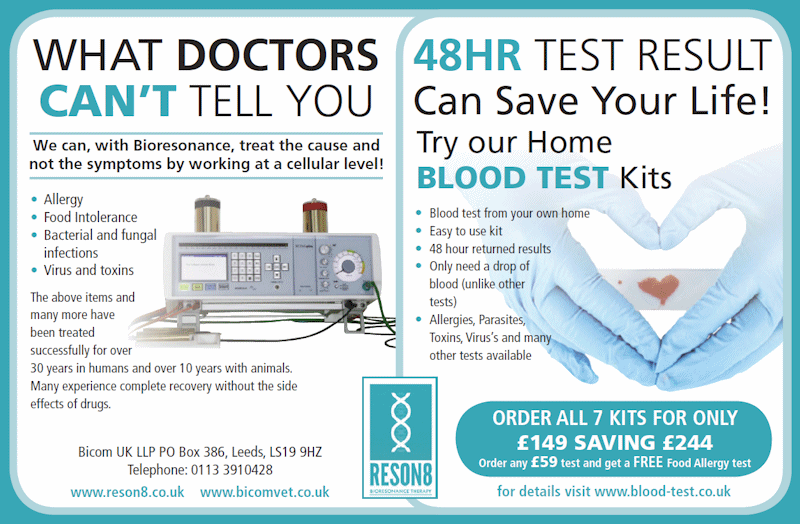 The list of conditions this device from Bicom can treat is long. With that and their cheap, 48-hour home blood test kit that only requires a drop of your blood, it's a wonder every hospital doesn't have one, particularly since Bicom claim many of these diseases have been treated successfully for over 30 years in humans. It must be a well-kept secret.
The list of conditions this device from Bicom can treat is long. With that and their cheap, 48-hour home blood test kit that only requires a drop of your blood, it's a wonder every hospital doesn't have one, particularly since Bicom claim many of these diseases have been treated successfully for over 30 years in humans. It must be a well-kept secret.
A well-kept secret that Bicom tells everyone about on their website. On multiple websites in fact. On the World-Wide Web. And trains people to use it in the twelve UK training courses they have scheduled for this year. Maybe we all need to tell our GP about it.
But maybe doctors already know, but don't tell you for some reason?
What Doctors Don't Tell You
Alternatively, there may be very good reasons why doctors don't tell you about this miraculous machine and the blood tests.
We came across an advert for the Bicom device and the home blood test kits in the September 2014 issue of What Doctors Don’t Tell You (WDDTY).
We wondered whether the advertiser would be able to substantiate the claims made in the ad and on his websites: www.reson8.uk.com and www.blood-test.co.uk related to the ad and another one owned by the same company, www.ebolatreatments.com so we submitted a complaint to the Advertising Standards Authority.
Their adjudication is published today, upheld on all six points.
The ad in WDDTY said:
WHAT DOCTORS CAN'T TELL YOU We can, with Bioresonance, treat the cause and not the symptoms by working at cellular level! Allergy Food Intolerance Bacterial and fungal infections Virus and toxins The above items and many more have been treated successfully for over 30 years in humans and over 10 years with animals. Many experience complete recovery without the side effects of drugs…48HR TEST RESULT Can Save Your Life! Try our Home BLOOD TEST Kits Blood test from own home Easy to use kit 48 hour returned results Only need a drop of blood (unlike other tests) Allergies, Parasites, Toxins, Virus's [sic] and many other tests available.
For the www.reson8.uk.com website, the ASA quoted some of the claims and said:
"Disorders Hayfever & Allergies Cancer Depression Smoking Weight Gain Electronic Smog Asthma Digestive Skin problems Gastro-intestinal diseases Auto Immune diseases Infectious diseases Neurological disorders Dental Pain Sports Injury Migraine". A video on the website gave details about how the treatment worked and featured individuals, some of which were introduced as medical professionals, talking about how the treatment had helped a number of conditions including gut problems, irritable bowel syndrome, Crohn's disease, allergies, neurodermatitis or ADHD and dental purposes. On a page, entitled "Bio-resonance treatment options for Ebola", text stated, "BICOM bioresonance therapy has been able to help many patients with just three or four sessions. Could this proven method of testing and treatment help people with Ebola and disable the virus early? Lets [sic] hope so…
Claims on www.blood-test.co.uk stated:
Home Blood Testing… Allergy testing in the UK has never been easier…During the testing, the frequencies of most known substances are measured against your blood sample. Upon detection, frequency patterns that match emit a "resonance" which is then recorded. The substances which contribute to your stress, or are likely to cause stress, or that may have caused stress in the past are rendered identifiable…For a fast, affordable, reliable and convenient home blood test service you need look no further than Blood Test UK…Order a free allergy test kit now
But perhaps most worryingly, on www.ebolatreatments.com:
Ebola Treatments and Testing: Give hope to those who need it most without medication! Bioresonance has not been tested or verified with the Ebola Virus, however it has been very successful with other hemorrhagic virus's [sic]. It is hoped that medical institutions in affected areas can apply our treatment protocols listed on this site and register their successful outcomes. We only use Class 2A medical devices such as the Bicom Optima which also is CE approved.
Further text on a page entitled Vaccine stated:
Ebola Vaccine No proven vaccine currently exists but…A bioresonance company in Australia has had some success preparing travellers against other pathogens like Bird Flu, Dengue, Japanese Encephalitis, Yellow Fever etc, in fact many pathogens can be used in a single tablet. The pathogenic frequencies are inverted and stored in pill form and taken during the trip.
Just to repeat those last few words:
The pathogenic frequencies are inverted and stored in pill form and taken during the trip.
You can see why we were concerned about these adverts — and we weren't the only ones. Someone else also complained, but of course, due to ASA confidentiality, we have no idea who that was.
The issues identified by the ASA were mostly concerned about whether the advertiser could substantiate the claims and whether the ads 'discouraged essential treatment for conditions for which medical supervision should be sought.' Like Ebola.
When asked by the ASA to substantiate the claims made, the advertiser, Bicom UK LLP, said:
…the claims were carefully worded, not making reference to the effectiveness of the treatment, and a disclaimer on their websites encouraged people to visit their doctor if they had a serious condition. They said that the listed disorders were being treated on a daily basis by 15,000 practitioners. They provided a 2009 press release following a German court case, which cited a decision not to impose an injunction on Bicom's claim to treat allergies, and an academic paper, which explored the electromagnetic effects on humans.
Needless to say, this 'evidence' did not satisfy the ASA's high standards. To understand how the ASA came to their decision to uphold all six points, please read their assessment of this 'evidence'.
The ASA decided the ads breached the CAP Code on a total of 18 counts and the advertiser was told:
The ads must not appear in their current form. We told Bicom to ensure that they held adequate substantiation for the efficacy claims made in their advertising and did not offer treatment or discourage essential treatment for conditions for which medical supervision should be sought.
Since the ad included claims about cancer, the ASA have referred the matter to Trading Standards (TS) for further investigation. It should be noted that TS have had a lot of success in prosecuting those making cancer claims. See Jo Brodie's blog post: Cancer Act 1939 convictions in the UK for full details.
Approved?
We noticed that Bicom had stated that:
We only use Class 2A medical devices such as the Bicom Optima which also is CE approved.
 This is a technical point about compliance with the EU Medical Devices Directive (MDD) 93/42/EEC and CE marking of a device. This CE marking is what is seen on many household items these days from electrical goods to toys and is a declaration by manufacturer or EU importer that the product meets the essential requirements of all applicable EU Directives, such as safety. For medical devices this is the MDD and there are several Classes, depending on the product and its function. Bicom are claiming their device is Class IIa, but we doubted this was correct and that they could substantiate this. The ASA took this up and told us:
This is a technical point about compliance with the EU Medical Devices Directive (MDD) 93/42/EEC and CE marking of a device. This CE marking is what is seen on many household items these days from electrical goods to toys and is a declaration by manufacturer or EU importer that the product meets the essential requirements of all applicable EU Directives, such as safety. For medical devices this is the MDD and there are several Classes, depending on the product and its function. Bicom are claiming their device is Class IIa, but we doubted this was correct and that they could substantiate this. The ASA took this up and told us:
We requested that they provided evidence to substantiate the claims “Class 2a” and “CE approved” appearing on www.ebolattreatments.com. Further to this, the advertiser provided some documentation to substantiate the claims. As we were not initially certain were the certifications were legitimate, we contacted the Medical Healthcare Regulatory Agency (MHRA) who confirmed that they were and, therefore, that the evidence did substantiate the claims.
However, the MHRA have decided to investigate the advertiser on the basis that they may not be using the device in the certified way. Unfortunately, due to statutory confidentiality restrictions, the MHRA will not be able to provide any further information about their investigation or update us on its progress.
Although the MHRA are investigating, this highlights two problems: how such a device ever came to be classified under IIa and the secrecy under which the MHRA operate.
Dealing with the first point, under the MDD, a manufacturer has to provide a dossier to a Notified Body to check everything is in order and it looks like the MHRA have checked this and decided the paper work was all present and correct. We believe there is a serious problem with how this works and the ease with which devices can appear to be compliant with the MDD with no evidence that they actually work. Possibly the worst example of this we have found was ear candles marked as Class I medical devices. This was a few years ago and the MHRA dealt with it, but this brings us on to the second issue: that of MHRA secrecy.
We don't know the details in this case, but the MHRA sometimes cite Article 20 of the MDD on confidentiality:
Without prejudice to the existing national provisions and practices on medical confidentiality, Member States shall ensure that all the Parties involved in the application of this Directive are bound to observe confidentiality with regard to all information obtained in carrying out their tasks.
There are certainly arguments to be made about treating possibly commercially sensitive information that the MHRA obtains from a manufacturer in the course of an investigation, but we believe that this cannot be interpreted as covering all information relating to a complaint or investigation, including whether a complaint had any merit, whether the MHRA investigated, what they found and concluded and what action they took or sanctions they imposed in order to fulfil their duty to enforce the MDD and protect the public.
This is certainly something we will return to at a later date.
 FirZone
FirZone
We didn't say much about it at the time, but we had also submitted a complaint about another advert in the September 2014 issue of What Doctors Don't Tell You. This one was informally resolved by the ASA and only the advertiser's name and brief details were published on the ASA's website on 5 November 2014.
The products being advertised were various models of sauna, with these claims:
Japanese studies have shown that Far Infrared Saunas are beneficial for arthritis, fibromyalgia, chronic fatigue, diabetes, insomnia, stress, psoriasis, chronic pain and can aid detoxification, weight loss, and fitness.
As with the Bicom WDDTY ad above, we also looked at this advertiser's website www.firzone.co.uk. We included some examples of the claims made on the website in our complaint, including:
Firzone Tourmaline Dome sauna with FIR heated mat (Cached)
Its application is suitable for slimming, detoxification, complementary cancer therapy (hyperthermia), cardio vascular fitness, treatment of aches & pains, healing, or as a pre massage treatment to relax muscles and increase blood flow.
Portable Infrared Sauna (large) (Cached)
Some of the reported uses and benefits of taking a FIR sauna include skin cleansing, detoxification, slimming, cardiovascular training, improving circulation, and promoting relaxation.
TourmalinePro with ceramic heater (Cached)
Health Benefits of Infrared Sauna
- Treat muscular aches and & sprains
- Improve circulation
- Promote weight loss
- Have healthy looking radiant skin
- Relax mind and body
- Enhance detoxification processes
- Improve cardiovascular conditioning and fitness levels
Portable Infrared Sauna Blanket (Cached)
Uses
- Slimming
- Detoxification
- Immunity
- Cancer
- Fatigue
- Treatment of aches and pains
- General fitness
- Relaxation
We don't know if the advertiser provided the evidence they mentioned in the ad or any other evidence they might have had, or whether they just agreed to remove the claims the ASA were unhappy about. All we know is that they removed these claims from their website: it's always interesting to see how an advertiser tries to substantiate claims, but removing the claims is still a good result.
Because of the cancer claims, we brought this to the attention of Trading Standards (via the Citizens Advice Bureau's webform) who investigated. Once the advertiser had removed the claims, TS closed the case.
More to come
We found other adverts in What Doctors Don't Tell You — and on those advertisers' websites — we were concerned about and submitted comprehensive complaints about those as well. They are still being investigated by the ASA, but we'll bring you the details when the outcomes are published.
It is however, very disappointing, despite the large number of previous adjudications and informally resolved cases about adverts published in WDDTY, that its publishers continue to carry ads making misleading claims and that some of their advertisers have been found to be publishing ads that are not only misleading, but are also irresponsible.
28 January 2015
Latest news
- "Undisputable evidence of scientific misconduct" by homeopaths
- Yet another bad year for homeopathy
- Nelsons Homeopathic Pharmacy #3
- Nelsons Homeopathic Pharmacy #2
- The Society of Homeopaths: failing to make the case for homeopathy
- The end of homeopathy on the NHS in Bristol?
- NHS Homeopathy: 20 years of decline
- The different faces of the Society of Homeopaths
- The growing pains of osteopaths
- Diluting misleading claims - ASA update
Most read
- Finding deleted and changed webpages
- About The Nightingale Collaboration
- How to find out who owns a website
- Advertising Standards Authority
- Rubbing salts into the wounds of homeopathy
- How to submit a complaint to the ASA
- The decline of homeopathy on the NHS
- Landmark decisions for homeopaths
- NHS Lanarkshire to end referrals to Glasgow Homeopathic Hospital
- Making a complaint
